P. Patel, A. Aranha, L. Cardozo
Wayne State University School of Medicine, Department of Internal Medicine, Division of Geriatrics, Detroit, Michigan 48201-2153, USA. Corresponding author: Pragnesh Patel M.D. Wayne State University School of Medicine, 5C, University Health Center, 4201 St Antoine Dr, Detroit MI 48201-2153, Phone: (313)577-5030, Fax: (313)745-4707, Email: pjpatel@med.wayne.edu
Abstract
Background: Pressure Ulcers (PU) have grown into a significant clinical and economic problem; as a consequence there is a need for a better understanding of predictors for PU development in Nursing Home (NH) Residents (NHR). Objective: To analyze the prevalence and predictors of PU among United States NHR population. Methods: The Center of Disease Control National Nursing Home Survey 2004 comprising 13,507 NHR from 1,152 United States NH facilities. Among them, 11,870 NHR, above age 65, having or not having Stage 1-4 PU, were analyzed. Results: NHR mean age was 84.2 years, 74.4% female, 89.4% white, 5.2% behavioral problems, 5.6% dementia and 10.3% stages 1-4 PU. Age of cohort was directly related to Activities of Daily Living (ADL) dependencies (p=0.001) and inversely to number of medications taken (p=0.001). Overall, the NHR with PU were slightly younger (p=0.021) with a shorter Length of Stay (LOS) (p=0.001), had skin wounds present (p=0.001), a higher ADL dependency (p=0.001), and number of medications taken (p=0.001). NHR without behavioral problems had a greater likelihood (p=0.021) of having PU. Conclusion: Age, Gender, Ethnicity, LOS at NH, presence of skin wounds, number of ADL dependencies and Medications taken were found to be good predictors of PU among NHR.
Key words: Nursing home residents, pressure ulcers, elderly.
Introduction
Pressure Ulcers (PU) are a common problem in Nursing Homes (NH) across the United States. A PU is defined as a localized injury to tissue overlying a bony prominence.(1, 2). The incidence and prevalence of PU has grown with aging and increase in the life span (3, 4). Long-term care residents are at a higher risk of developing PU because of a variety of risk factors that include immobility, malnutrition, incontinence and dementia (4). PU adversely affect the quality of life and is also associated with high cost (estimated at $3.3 billion per year in 2008) and high mortality rates (5, 6). About 60,000 U.S. patients die annually from PU complications.(7, 8). The Centers for Medicare and Medicaid Services in 2006 declared hospital-acquired stage III or stage IV pressure ulcers as adverse patient safety events, that could reasonably be prevented by the implementation of evidence-based prevention guidelines. As a result, the inpatient cost of hospital acquired Stage III and IV ulcers are not reimbursed.(9, 10). Similar reimbursement changes could in future affect Nursing homes. It is therefore important, to be able to predict the risk for PU development in NH Residents (NHR) and implement appropriate preventive measures (11). However, despite preventive measures, PU can still develop in NHR as a consequence of multiple co-morbidities and underlying risk factors. There is, therefore, a need to better define these predictors and institute appropriate preventive cost effective interventions to facilitate improvement in health and quality of life outcomes. The present study evaluates the risk factors associated with PU, in a large NHR survey, with a view to gain a better understanding of the risk factors that can predict PU development in NHR. To date, this is the largest survey that we are aware of.
Methods
Survey methodology: We accessed a data file survey from Centers for Disease Control and Prevention (CDC website) (12), and conducted an analysis of the Center of Disease Control’s National Nursing Home Survey (NNHS) 2004. This NNHS survey comprised of 13,507 NHR from 1,152 Nursing Home facilities, randomly selected within the United States. Among them, a total of 11,870 NHR, above the age of 65 years, were identified as either having or not having stage 1 to 4 PU as defined by National Pressure Ulcer Advisory Panel (2).To facilitate optimal use of all data in the NNHS, cases with variables having “Don’t Know” or “Not Ascertained” were scored as “Missing” for each of those case variables and all such cases were retained in analysis of other variables.
Statistical Analysis
The data were analyzed using SPSS for Windows version 22.0 (IBM SPSS Inc., Chicago, IL). For analysis, NHR were grouped by presence of PU (Stage 1-4 PU and No PU). Continuous data (e.g. age) of two groups were analyzed using t test, and categorical data (e.g., gender) were evaluated using χ2 test. Pearson correlation coefficients (r) were used for analysis of degree and direction of the associations between the continuous data. Results are presented as Mean (M) ± Standard Deviation (SD) or as number (n) and percentage (%). Statistical significance was established at P <0.05.
Results
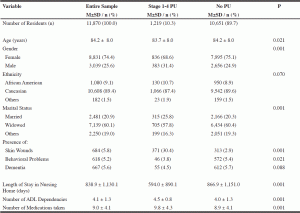
Sample characteristics of 11,870 NHR, partitioned by presence or absence of PU, are presented in Table 1. The NHR had a mean age of 84.2 years, 74.4% were females, 89.4% were White, the majority (81.0%) were either married (20.9%) or widowed (60.1%), and 7.2% were Veterans. A total of 5.2% had behavioral problems, 5.6% dementia, and 5.8% skin wounds. There were 10.3% with stages 1-4 PU, of which stage 2 was a majority (51.8%) and stage 1 a minority (9.7%), with other stages of PU recording 25.9% (stage 1) and 12.5% (stage 4).
Note: M=Mean; SD=Standard Deviation; PU=Pressure Ulcer; ADL=Activities of Daily Living.
Among the NHR, a larger proportion (11.7%) of the Veterans had PU. The age of the cohort was directly (positively) related to Activities of Daily Living (ADL) dependencies (p=0.001) and inversely (negatively) to number of medications taken (p=0.001). Overall, the NHR with PU were younger (p=0.021) with a lower Length of Stay (LOS) at the NH (p=0.048), had a higher ADL dependency (p=0.001) and number of medications taken (p=0.001). Although, no such relationship was observed with NHR having dementia, the NHR free of behavioral problems had a greater likelihood (p=0.021) of having PU. There were differences in the prevalence of PU among genders. The NH males were younger than the females (p=0.001) but had a higher proportion (12.6% versus 9.5%, p=0.001) of PU as compared to females. Although ethnic differences were not clearly evident in the prevalence of PU (p=0.070) among NHR, African Americans in the NH had a higher proportion (12.0%) of PU as compared to White (10.0%). A smaller proportion of NHR with Behavioral problems (3.8% versus 5.4%, p=0.021) and Dementia (4.5% versus 5.7%, p=0.088) had PU. Also, the NHR with skin wounds had a much higher proportion (30.4% versus 2.9%, p=0.001) of PU than those without any skin wounds.
Discussion
To our knowledge this is the first study to analyze a large sample size of NNHS data for PU in NHR. Among the risk factors examined, age, gender, ethnicity, length of stay at the nursing home, presence of skin wounds, total number of ADL dependencies and medications taken were associated with higher risk of PU in NHR. Of note, although the presence of dementia did not increase the risk of PU in this study, the presence of behavioral problems was in fact associated with a reduced risk of PU development.
The common risk factors for PU are immobility, urine and stool incontinence, malnutrition, dementia, pressure, shear and friction forces. Prevention of PU remains the best management strategy. For predicting the risk of PU, validated scales for PU formation like Braden scale can be used (13, 14). Braden scale is the most widely used risk assessment tool in most NH settings (1, 15). The NNHS sample had some of the same risk factors identified in the Braden scale.
As with previous studies (1, 11, 16), stage 2 PU were more common. Our data analysis also revealed that pressure ulcers were more likely to be found in patients who were younger, had greater dependency for ADL, took a higher number of medications and had a shorter NH stay as compared to patients with longer stays. The latter result is contrary to the findings of a previous study by Berlowtiz and coworkers (17), that showed the newly admitted resident to the long-term care facility having a lower risk of developing a new PU compared to existing residents. However, our findings are consistent with the study by Bergstrom and associates (18), that revealed that the majority of residents who developed a PU did so either in the first 3 weeks after admission to the facility or were likely to have a PU on admission to NH (19, 20). Additionally, the patients who were taking 9 or more medications in this study were likely to have a higher stage PU compared to those taking fewer medications. Although previous studies have not observed any such association between number of medications and PU, one study did note that taking antidepressants may lower the risk of PU by improving the mood (21, 22). A possible explanation for the increased incidence and higher stages of PU in residents taking a greater number of medications may be indicative of the number of co-morbidities as well as the severity of those illnesses.
African Americans were also more likely to have PU based on NNHS data compared to Whites. This may be partly related to the site and type of the Nursing facility and whether or not the appropriate PU risk assessment programs were utilized at the participating facilities (23). Furthermore, those with any type of skin wound had a greater likelihood of having a PU based on our NNHS analysis. This might be due to the underlying edema surrounding the wound as well as other co-morbidities, such as inflammatory conditions, leading to poor tissue oxygenation which is conducive to the development of PU.
Similar to other studies (24-26), there was a direct relationship with PU stage and ADL dependency. PU have been known to heal slowly, especially if a resident is ADL dependent, indicating a lower activity level and conversely heal faster if a resident with PU experiences bouts of agitation (27). Likewise, in our study there was a inverse correlation between behavioral problems and presence or risk of PU, perhaps, indicating that agitated residents who are prone to move around and be more active have a reduced risk of PU.
Our study has several limitations. It did not take into account other risk factors identified in the validated Braden or Norton scales (28) that are usually associated with a greater risk of PU development. For example, the nutritional state that is often a significant risk factor for PU development was not documented in this survey. While ADLs were a factor in PU development, the role of immobility (29), incontinence (30), and impaired mental status (31), were not studied. Specifically, sensory perception and the ability of the NHR to participate in their own care are of importance. Finally, this study does not address other factors related to the nursing home environment of care, such as the competency of the providers to recognize and grade PU, barriers to implementation of preventive care and therapeutic modalities. The use of pressure redistribution mattresses were also not part of the NNHS study (32). However, this large NHR survey validates some of the risk factors identified in Braden and Norten scales. It also identifies other factors of importance in the genesis of PU.
Conclusion
This study has identified several risk factors that are associated with PU in NHR. The residents’ age, gender, ethnicity, LOS at the NH, presence of skin wounds, the total number of ADL dependencies and medications taken were found to be good predictors of PU in NHR. The presence of dementia did not increase the risk of PU among NHR, however, the presence of behavioral problems was associated with a reduced risk of PU development. Identification of these PU predictors should enable the NH implement PU-prevention and treatment protocols for at-risk NHR.
Source of Funding: None.
Author Disclosures: The authors report no relationship or financial interest with any entity that would pose a conflict of interest with the subject matter of this article.
Ethical standards: The National Nursing Home Survey 2004 data was collected by CDC, a US federal government agency, using the highest approved Ethical Standards.
References
1. European Pressure Ulcer Advisory Panel and National Pressure Ulcer Advisory Panel. Treatment of pressure ulcers: quick reference guide. http://www.epuap.org/wp-content/uploads/2010/10/Quick-Reference-Guide-DIGITAL-NPUAP-EPUAP-PPPIA-16Oct2014.pdf. Accessed December 4, 2014.
2. NPUAP pressure ulcer staging and 2007 update. National Pressure Ulcer Advisory Panel 2007. http://www.npuap.org/resources/educational-and-clinical-resources/npuap-pressure-ulcer-stagescategories. Accessed December 4, 2014.
3. Lyder CH, Ayello EA. Annual checkup: the CMS pressure ulcer present-on-admission indicator. Adv Skin Wound Care 2009; 22:476-484.
4. Pham B, Stern A, Chen W, et al. Preventing pressure ulcers in long-term care. Arch Intern Med 2011;171(20):1839-1847.
5. Thein HH, Gomes T, Krahn MD, Wodchis WP. Health status utilities and the impact of pressure ulcers in long-term care residents in Ontario. Wual Life Res 2010;19(1):81-89.
6. Vand Den Bos J, Rustagi K et.al. The $17.1 billion problem: the annual cost of measurable medical errors. Health Aff (Millwood) 2011;30(4):596-603.
7. Lyder CH. The benefits of a multi-disciplinary approach to the prevention and treatment of pressure ulcers. Published Infection Control Today, August 2011. http://www.infectioncontroltoday.com/news/2011/08/the-benefits-of-a-multi-disciplinary-approach-to-the-prevention-and-treatment-of-pressure-ulcers.aspx. Accessed December 4, 2014.
8. Sullivan N, Schoelles KM. Preventing in-facility pressure ulcers as a patient safety strategy. Ann Intern Med 2014; 158:410-416.
9. Centers for Medicare and Medicaid Services. Eliminating serious, preventable and costly medical errors–never events. Centers for Medicare and Medicaid Services Web site. http://www.cms.hhs.gov/apps/media/press/releas. Published May 18, 2006. Accessed December 3, 2014.
10. Centers for Medicare and Medicaid Services. CMS proposes additions to hospital-acquired conditions for fiscal year 2009. http://www.cms.gov/apps/media/fact_sheets.asp. Published April 14, 2008. Accessed December 4, 2014.
11. Cox J. Predictors of pressure ulcers in adult critical care patients. Am J Crit Care 2011; 20(5):364-375.
12. National Nursing Home Survey.www.cdc.gov/nnhs/nnhs/questionannarires.htm. Accessed on December 20, 2014.
13. Bergstrom N, Braden B, Kemp M, Champagne M, Ruby E. Predicting pressure ulcer risk: a multisite study of the predictive validity of the Braden Scale. Nurs Res 1998;47(5):261-269.
14. Pancorbo-Hidalgo PL, Garcia-Fernandez FP, Lopez-Medina IM, Alvarez-Nieto C. Risk assessment scales for pressure ulcer prevention: a systematic review. J Adv Nurs 2006;54(1):94-110.
15. Bergstrom N, Braden B, Laguzza A, Holman V. The Braden Scale for predicting pressure sore risk. Nurs Res 1987;36(4):205-210.
16. Smith DM. Pressure ulcers in the nursing home. Ann Intern Med 1995;123:433-442.
17. Berlowitz DR, Ash AS, Brandeis GH, Brand HK, Halpern JL, Moskowitz MA. Rating long-term care facilities on pressure ulcer development: importance of case-mix adjustment. Ann Intern Med 1996;124:557-563.
18. Bergstrom N, Braden B. A prospective study of pressure sore risk among institutionalized elderly. J Am Geriatr Soc 1992;40:747-758.
19. Ek AC, Unosson M, Larrson J, Von Schenck H, Bjurulf P. The development and healing of pressure sores related to the nutritional state. Clin Nutr 1991;10(5):245-250.
20. Horn SD, Bender SA, Bergstrom N, et al. Description of the national pressure ulcer long-term care study. J Am Geriatr Soc 2002;50(11):1816-1825.
21. Brandeis GH, Ooi WL, Hossain M, Morris JN, Lipsitz LA. A longitudinal study of risk factors associated with the formation of pressure ulcers in nursing homes. J Am Geriatr Soc 1994;42(4):388-393.
22. Horn SD, Bender SA, Ferguson ML, et al. The national pressure ulcer long-term care study: pressure ulcer development in long-term care residents. J Am Geriatr Soc 2004;52(3):359-367.
23. Li Y, Yin J, Cai X, Temkin-Greener J, Mukamel DB. Association of race and sites of care with pressure ulcers in high-risk nursing home residents. JAMA 2011;306(2):179-186.
24. Aydin G, Mucuk S. The evaluation of daily living activities, pressure sores and risk factors. Rehabil Nurs. 2015 Mar-Apr;40(2):84-91.
25. Lindgren M, Unosson M, Fredrickson M, Ek A. Immobility — a major risk factor for development of pressure ulcers among adult hospitalized patients: a prospective study. Scand J Caring Sci. 2004;18(1):57-64.
26. Bergquist-Beringer S, Gajewski BJ. Outcome and assessment information set data that predict pressure ulcer development in older adult home health patients. Adv Skin Wound Care. 2011 Sep;24(9):404-414.
27. Bergstrom N, Smout R, Horn S, Spector W, Hartz A, Limcangco MR. Stage 2 pressure ulcer healing in nursing homes. J Am Geriatr Soc 2008; 56(7):1252-1258.
28. Norton D, McLaren R, Exton-Smith AN. An investigation of geriatric nursing problems in hospitals. London: National Corporation for the Care of Old People; 1975.
29. Baumgarten M, Margolis DJ, Localio AR, et al. Pressure ulcers among elderly patients early in hospital stay. J Gerontol A Biol Sci Med Sci 2006; 61(7):749-754.
30. Maklebust J, Magnan MA. Risk factors associated with having a pressure ulcer: A secondary data analysis. Adv Wound Care 1994;7(6):25-28.
31. Sullivan N, Karen M, Schoelles KM. Preventing in-facility pressure ulcers as a patient safety strategy. Ann Intern Med 2013; 158:410-416.
32. Black J. Pressure ulcer prevention and management: A dire need for good science. Ann Intern Med 2015; 162:387-388.