C. Ruggiero1, M. Baroni1, E. Zengarini1, G. Dell’Aquila2, A. Cherubini2, V. Boccardi1, F. Lattanzio3, P. Mecocci1
1. Institute of Gerontology and Geriatrics, Department of Medicine, University of Perugia, Perugia, Italy; 2. Geriatrics and Geriatric Emergency Care,IRCCS-INRCA, Ancona, Italy; 3. Scientific Direction, Italian National Research Center on Aging, Ancona, Italy. Corresponding author: Carmelinda Ruggiero, MD, PhD, Department of Medicine Institute of Gerontology and Geriatrics, Ospedale S. Maria della Misericordia P.le Menghini,1 06123, Perugia, Italy, Tel. +39-075-578-3564 Fax. +39-075-5783878, E-mail: carmelinda.ruggiero@unipg.it
Abstract
Background: Identifying older individual at risk for fragility fractures is a priority among healthcare providers. However, the prevalence of osteoporosis, fragility fractures and the prescription of antifracture drugs among Nursing Home (NH) residents is almost ignored. The aim of this study is to describe the prevalence of osteoporosis and fragility fractures, and the main correlates of antifracture drug prescriptions among NH residents. Design: Observational multicentre prospective study. Setting: Nursing homes and assisted living facilities. Participants: 1995 NH residents aged 60 and older participating to the U.LI.S.S.E. (Un Link Informatico sui Servizi Sanitari Esistenti per l’Anziano) project were evaluated using a standardized comprehensive geriatric assessment instrument (Resident Assessment Instrument Minimum Data Set – RAI-MDS). Results: 256 (13%) persons (age 84.5±7.62 years) have diagnosis of osteoporosis without any fracture; 355 (17%) had previous fragility hip fracture with one out of three reporting a diagnosis of osteoporosis. An antifracture treatment is prescribed to 5.5% (n:111) of eligible persons: 18% of osteoporotic and 11% of hip fracture residents, respectively. Independent of age, gender, BMI and disability, subjects affected by osteoporosis are more likely to receive antifracture drugs (β=0.17, SE: 0.04; p: <0.0001),but not those with previous hip fracture. The probability to receive treatments decreases when dementia co-occurs (β=-0.07, SE: 0.03; p:0.05). Residents managed according with RAI-MDS show higher probability to receive appropriate antifracture treatment (β= 0.07, SE: 0.03; p: 0.01). Conclusion: NH residents at high risk for fragility fractures receive suboptimal care. Residents with a history of hip fracture or dementia are less likely to be treated, while those managed according to the geriatric comprehensive approach are more likely to receive adequate care.
Key words: nursing homes, antifracture drugs, fragility fracture, observational study, care gap.
Introduction
Osteoporosis is a skeletal disorder characterized by compromised bone strength, which leads to fragility fractures (1). Osteoporotic fractures are particularly common in frail older individuals. Chronic disease states and polypharmacy further contribute to deleterious effects on bone health(2). Within the elderly population, those living in nursing homes (NH) generally comprise the oldest and most frail subgroup of individuals (3). Epidemiological studies conducted in U.S. indicate that osteoporosis is prevalent among NH residents, with approximately 50% of men and 64-90% of women meeting the World Health Organization criteria for central or peripheral DXA (4,5). The rate of hip fracture per 100 NH residents per year has been estimated between 3.7 and 5.0 which reflects a fracture risk 2.5-10 times greater than that of community-dwelling older individuals (5). Limited data exists regarding the prevalence of osteoporosis and fragility fractures among European NH residents. According to some authors, the rates of hip fractures range from 8% in Netherlands, to 10% in Finland, 18% in Italy and 21% in Austria (6,7). The prevalence of osteoporosis and vertebral fractures has also been shown to be 52% and 36%, respectively, among oldest NH residents (mean age, 85.9±0.6 years) living in Geneva, Switzerland. Additionally, the ten-year fracture probability as assessed by the FRAX tool was 27% and 15% for major fractures and hip fractures, respectively (8).With hip fracture comes the associated risk for further disability, hospitalization, and healthcare utilization. Therefore, hip fracture reduction is an essential concern in healthcare policy (9-13).
Falls and low bone mineral density (BMD) are the main determinants of hip fractures among NH residents and, therefore, are the key targets of interventions. The effect of fall prevention on hip fracture reduction remains unclear, especially among NH residents (14). In addition, they seldom receive antiosteoporotic drug treatments (i.e. vitamin D, calcium and bisphosphonates) although strong evidence supporting the improvement of the BMD and the reduced risk of fracture (15,1 6). Fracture risk is often underestimated among NH residents. Therefore, osteoprotective agents are generally under-prescribed, with rates as low as 20% in some cases (17). Due to a lack of data in the literature, the goals of this study were to assess the prevalence of osteoporosis and hip fragility fractures among NH residents, and the rate of antifracture drug use among participants in the Un Link Informatico sui Servizi Sanitari Esistenti per l’anziano – A computerized network on healthcare services for the elderly (ULISSE) study.
Methods
Study sample
The study sample consisted of older individuals participating in the ULISSE project, an observational multicentre, prospective one-year study investigating the characteristics of older patients and the quality of care provided to them in acute, home, and NH care settings. This study was approved by the ethics committee of The Catholic University of Rome. Briefly, in February 2004, 1775 residents aged 60 years or older, living in 31 NHs distributed across Italy were enrolled and were followed until August 2007. In each instance where a participant was discharged or died, a new participant replaced him or her.In addition to the 1775 residents enrolled at baseline, 202 residents were enrolled in 2005, 26 subjects in 2006, and 16 subjects in 2007, for a total of 2019 subjects. 1995 subjects with complete data and information for the purpose of the study were included in the analysis. Details about the methods and procedures of the ULISSE project have been previously published (18).
Data collection
Italian NH residents participating in the ULISSE project underwent a standardized comprehensive evaluation protocol using the Italian version of the Resident Assessment Instrument Minimum Data Set (RAI-MDS) for NH care (19). The RAI-MDS, currently recommended by the Italian Geriatrics Society, has been previously validated and is currently in use among Italian nursing homes (20). At baseline, participants underwent a comprehensive geriatric assessment of functional and cognitive status evaluated by means of the seven point MDS activities of daily living hierarchy (21) and the 6 point MDS cognitive performance scale (CPS), respectively (22). Additional standardized geriatric assessments were used, including a full list of medical diagnoses coded according to the ICD-9 system (23), detailed data on long-term medications used in the past 90 days, and the Cumulative Illness Rating Scale (CIRS) to quantify comorbidity (24), an ad-hoc designed questionnaire to collect data on NH characteristics. For the purpose of the present study, participants with osteoporosis were identified based on a diagnosis reported in the dedicated section of the RAI-MDS for NH. Fragility fractures were identified as those occurring at typical sites (i.e. wrist, lumbar spine, hip, humerus, radius and ulna) after the age of 60 years. Fractures secondary to traumatic accidents or cancer were excluded. Among the participants with fragility fractures, those reporting a hip fracture were included in the “Hip Fracture” independent of the presence of any other fracture, while those with any other fracture type were included in the “Other Fracture” group. Data on prescription medications were obtained from medical records. Drug information, such as name, form, starting time, dosage, and frequency of administration, were recorded for each resident. Drugs were coded using the Anatomical and Therapeutic Classification system (25). All participants included in the study were on long-term drug treatment, which was defined as the continuation of the same drug therapy for 6 months prior to baseline evaluation.
Statistical analysis
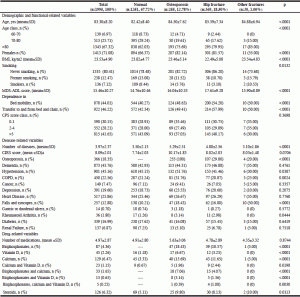
Table I shows the main characteristics of the NH residents participating in the ULISSE project. Data are presented for the entire sample and classified as “Normal”, subjects without diagnosis of osteoporosis and/or previous fracture, as “Osteoporosis”, those with diagnosis of osteoporosis without previous fracture, and “Hip fracture”, those with previous hip fracture independent of previous diagnosis of osteoporosis, and “Other fracture”, subjects with any other fracture and independent of previous diagnosis of osteoporosis. Table 2 reports the characteristics of the participants according to on-going anti-osteoporotic treatment. In both Table 1 and Table 2, variables with normal distribution were summarized by mean and standard deviation, or by proportion and percentage. Statistical differences were tested using Student’s t-test, ANOVA with Bonferroni correction, or Chi-square test as appropriate. Correlates of anti-osteoporotic drug treatment among NH residents were identified based on a multivariate regression model using GLM procedure (Table 3). A probability value of <0.05 was considered statistically significant. All statistical analyses were conducted using SAS version 9.13 (SAS Institute, Cary, NC, USA).
Data are presented as mean±standard deviation or proportion and percentages, as appropriate. Statistical differences are tested using the ANOVA analysis of variance or Chi-square test, as appropriate; MDS-ADL score: MDS-ADL Long Form Scale (score 0-28); CPS: Cognitive Performance Scale; CIRS: Cumulative Illness Rating Scale; COPD: Chronic Obstructive Pulmonary Disease; a. Log-transformed values were then back-transformed for data presentation.
Results
The baseline characteristics of the study sample (n=1995) are presented in Table I. The majority of participants were women (n=1413, 71%), aged 80 years or older (n=1343, 67%), with a mean BMI of 23.53kg/m2 and mean MDS-ADL score of 15.46 (median=17; interquartile range 5-25). A large portion of subjects were dependent on bed mobility (n=878, 44.01%) and on bed-to-chair transfers (n=922, 46.22%), and had a severe level of CPS-based cognitive impairment (n=815, 41.65%), independent of a ICD-9 based diagnosis of dementia. Residents had a high level of multimorbidity, as shown by the number of diseases (3.97±2.37), and the CIRS score (8.09±2.01), and the average number of drugs (4.97±2.87). The prevalence of diseases and conditions affecting NH residents are listed in Table 1. After hypertension (45.16%), dementia (43.76%) was the most prevalent disease, followed by heart disease (25.86%), COPD (22.56%), depression (19.60%), diabetes (16.99%), falls and syncope (12.88%). Concerning the prevalence of bone-related diseases, 366 (18.35%) residents had osteoporosis (82.14% women; 75.69% octogenarians), 369 (18.50%) had at least one fragility hip fracture (82.14% women and 79.95% octogenarians), and 20 (1.00%) had other fractures. Many residents with hip fracture (262; 71.00%) did not have a clinical diagnosis of osteoporosis. Including hip and other fragility fractures, osteoporosis was the third most common chronic diseases among Italian NH residents in this study, with a mean prevalence of 32.28% (Table 1).
Compared to normal residents, those with osteoporosis and hip fracture were older (84.50±7.62 and 85.59±7,respectively), had a higher MDS-ADL disability (16.04±10.33 and 17.61±9.28, respectively), including dependence on bed mobility and bed-to-chair transfers, higher multimorbility (5.29±2.51 and 4.80±2.56, respectively) despite a similar rate of polypharmacy (5.63±3.06 and 4.78+2.89, respectively). Residents grouped as osteoporotic or hip fractured had dementia (113; 44.31% and 173; 46.88%), falls and syncope(47;18.43% and 62;16.80%) as prevalent comorbid conditions. Concerning antifracture treatment, 87(4.36%) residents received specific drugs, with 129 (6.47%) and 45 (2.26%) on calcium or vitamin D supplementation, and 5 (0.25%) receiving antiresorptive drug plus supplements of calcium and vitamin D (Table I). Concerning the pattern of antiresorptive drug use, the majority of residents receive clodronate (n=61, 70.11%), followed by alendronate (n=17, 19.54%), risedronate (n=7, 8.05%), neridronate (n=1, 1.15%) and etidronate (n=1, 1.15%). 5 (0.8%) of 624 (31.27%) residents with a clinical diagnosis of osteoporosis and/or related hip fragility fractures received a complete osteoprotective therapy, while 86 (13.78%) residents were treated with bisphosphonates alone, 29 (4.64%) with vitamin D, and 83 (13.30%) calcium supplement alone.
Table 2 shows the characteristics of NH residents categorized according to antiosteoporotic drug use. Compared to those untreated, the majority of residents taking these drugs had a clinical diagnosis of osteoporosis (85.06%), and approximately one out of two had hip fracture (44.83%). The majority of residents with CPS-based severe cognitive impairment (n=222, 92.8%) and dementia (n=259, 90.0%) treatment despite their diagnosis (Table 2).
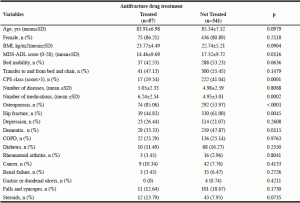
Table 2 Characteristics of the Italian NH Residents Participating in the U.L.I.S.S.E. Project according to Antifracture Drug Treatment
Independent of age, sex, BMI and disability score, residents affected by osteoporosis were more likely to receive an antiosteoporotic drug (β=0.20, SE=0.04, p<0.0001), while there was no association between hip fracture and treatment (β=0.20, SE=0.04, p<0.0001)(Table 3, Model 1). Residents with severe CPS-based cognitive impairment (β=-0.08, SE=0.03, p=0.0218), not those having a diagnosis of dementia in the clinical chart (β=-0.04, SE=0.03, p=0.1457), and residents with history of falls and syncope(β=-0.08, SE=0.03; p=0.0388) were less likely to receive treatment (Table 3, Model 2 and 3). Residents clinically managed based on a multidimensional geriatric instrument, i.e. RAI-MDS, were more likely to receive treatment (β: 0.07, SE: 0.03; p: 0.0134), especially when a history of falls, syncope, severe cognitive impairment were detected (Table 3, Model 4).
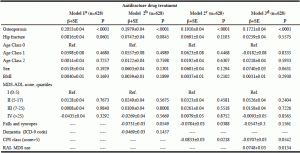
Table 3 Correlates of Antifracture Drug Treatment among the Italian NH Residents Participating in the U.L.I.S.S.E. Project
a. osteoporosis, hip fracture, age classes, sex, BMI, MDS-ADL score quartiles; b. osteoporosis, hip fracture, age classes, sex, BMI, MDS-ADL score quartiles, falls/syncopes, ICD-9 diagnosis of dementia; c. osteoporosis, hip fracture, age classes, sex, BMI, MDS-ADL score quartiles, falls/syncopes, high degree of cognitive impairment CPS-based; d. osteoporosis, hip fracture, age classes, sex, BMI, MDS-ADL score quartiles, falls/syncopes, high degree of cognitive impairment CPS-based, RAI-MDS use in clinical management
Discussion
This study investigated the prevalence of osteoporosis, fragility fractures and osteoprotective drug therapy in a large sample of elderly Italian NH residents. We found that osteoporosis is under-reported and under-diagnosed, even when fragility fractures were sustained. Furthermore, this study revealed a trend of under-treatment of osteoporosis among NH residents, even once clinical diagnosis is established. These data further support the notion that osteoporosis and fragility fractures are neglected conditions among residents of Italian NHs. Our sample revealed an osteoporosis prevalence rate of approximately 18%, which is consistent with the trend among European NHs (8-21%) (6,7), but far lower than that of USA NHs (70-85%) (2,4,5). After reviewing patient medical records, we found that osteoporosis and related-fractures were the third most common disease affecting Italian NH residents in this sample. Furthermore, Italian NH residents have high rate of condition-specific undertreatment. Only 2 out of 5 patients with a clinical diagnosis of osteoporosis and 1 out of 4 persons with fragility fractures received at least one osteoprotective drug, and only 0.39% and 1.08% received calcium or vitamin D plus an antiosteoporotic drug, respectively. The majority of treated residents received clodronate, a bisphosphonate with the poorest level of evidence for anti-hip fracture efficacy (26).
Our study adds to the literature by confirming that too few osteoporotic patients receive treatment before and after fracture, despite the availability of effective medications. Consistent with previous data, the likelihood of bisphosphonate prescription increases among NH patients with greater level of multimorbidity or polypharmacy (16, 17). Residents affected by osteoporosis and/or hip fracture have a higher likelihood of receiving antifracture medications, independent of their ability to ambulate (13, 17). Although the inability to walk could be considered a justification for exclusion of antiresorptive treatment, the probability of treatment was not modified by the inability of NH residents to move and stand-up. Additionally, NH residents with osteoporosis and/or hip fracture co-occurring with dementia were less likely to receive bisphosphonates. This observation deserves more attention considering that osteoporosis and dementia are commonly comorbid diseases in older NH residents, who are at highest risk for falls and hip fracture (27-29).
The reasons for these confounding medical practices remain unknown. Guidelines specific to NH patients continue to strongly recommend specific pharmacological treatment, in addition to calcium and vitamin D supplementation, especially in those patients with previous fractures (30). Concerns regarding tolerability, adverse drug effects, and polypharmacy likely continue to discourage physician prescription of antifracture drugs to this frail elderly population. This is especially true regarding patients with dementia diagnoses, which reduces drug compliance, for example in the case of bisphosphonates, which need to be taken on an empty stomach and without food for about an hour. Though this could be less of a problem with the assistance of nurses. In addition,widely available osteoporosis medications, such as parenteral amino-bisphosphonate (i.e. zoledronic acid), are more easily administered, have fewer compliance issues, and strong evidence of efficacy. Although there is an abundance of evidence indicating the efficacy of antiosteoporotic drugs in NH patients, these individuals are generally excluded from randomized controlled trials (31), which leaves the risk-benefit ratio and cost-effectiveness unresolved in this population. The lower rate of antiosteoporotic drug use in NH residents with dementia may indicate that osteoporosis is often unrecognized in elderly individuals with dementia until a fracture occurs. Interestingly, the management of the NH residents based on RAI-MDS increases the probability of receiving antifracture treatment. Therefore these findings suggest that an implementation of use of osteoporosis drugs could be achieved by providing an adequate care and optimizing the management of chronic diseases and polypharmacy in nursing home residents.
We acknowledge certain limitations to the current the study. The study was not specifically developed to identify the prevalence of bone disease and related treatment. This is a secondary analysis of the data. The nature of the study limits our ability to report pertinent data, including information on BMD scan, vitamin D levels, morphometric vertebral fractures, and treatment duration. However, the study does have several strengths: (a) the drug information was collected from the medical records and charts of each resident and reflects the drugs effectively administered; (b) the findings of this study are generalizable, since the data were collected from a wide national sample of NH residents; (c) to our knowledge, this is the first study investigating the correlates of antifracture drugs administration among Italian NH residents.
In conclusion, though NH residents are at high risk for falls and fracture, their diagnosis of osteoporosis is generally under-recognized or affected by previous major fragility fracture. They do not receive antifracture treatment, especially when dementia co-occurs, independent of their degree of disability. The clinical management of NH residents based on comprehensive care may help physicians to recognize persons who deserve antifracture treatments. These findings highlight the urgent need for intervention trials to test strategies that address the efficacy, tolerability, and cost-effectiveness of antifracture medications in high-risk NH patients.
Acknowledgements: The U.L.I.S.S.E. project was supported by the Italian Ministry of Health and Pfizer SrL Italy. The funding public agency and the private sponsor had no role in the design and conduct of the study, in the collection, analysis, and interpretation of the data; or in the preparation, review, or approval of the manuscript. Carmelinda Ruggiero, Marta Baroni, Elisa Zengarini, Giuseppina Dell’Aquila, Antonio Cherubini, Virginia Boccardi, Fabrizia Lattanzio, Patrizia Mecocci declare no conflict of interest. The research protocol was approved by the ethics committees and that written consent was obtained from all participants.
Conflict of interest: The funding public agency and the private sponsor had no role in the design and conduct of the study, in the collection, analysis, and interpretation of the data; or in the preparation, review, or approval of the manuscript. Carmelinda Ruggiero, Marta Baroni, Elisa Zengarini, Giuseppina Dell’Aquila, Antonio Cherubini, Virginia Boccardi, Fabrizia Lattanzio, Patrizia Mecocci declare no conflict of interest.
Ethical Standard: The research protocol was approved by the ethics committee and the written consent was obtained from all participants.
References
1. NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy Osteoporosis prevention, diagnosis, and therapy. JAMA 2001;285:785-795
2. Zarowitz BJ, Cheng LI, Allen C, O’Shea T, Stolshek B. Osteoporosis prevalence and characteristics of treated and untreated nursing home residents with osteoporosis. J Am Med Dir Assoc 2015;16:341-348.
3. Ruggiero C, Dell’Aquila G, Gasperini B, et al. Potentially inappropriate drug prescriptions and risk of hospitalization among Italian older nursing home residents: the U.L.I.S.S.E. project. Drugs Aging 2010;27:747-758
4. Zimmerman SI, Girman CJ, Buie VC et al. The prevalence of osteoporosis in nursing home residents. Osteoporos Int 1999;9:151-157
5. Chandler JM, Zimmerman SI, Girman CJ et al. Low bone mineral density and risk of fracture in white female nursing home residents. JAMA 2000;284:972-977
6. Achterberg WP, Gambassi G, Finne-Soveri H et al. Pain in European long-term care facilities: cross-national study in Finland, Italy and the Netherlands. Pain 2009;148:70-77
7. Pietschmann P, Azizi-Semrad U, Pils K, et al. Pharmacologic undertreatment of osteoporosis in Austrian nursing homes and senior’s residences. Wien Klin Wochenschr 2010;122:532-537
8. Rodondi A, Chevalley T, Rizzoli R. Prevalence of vertebral fracture in oldest old nursing home residents. Osteoporos Int 2012;23:2601-2606
9. Lee YK, Lee YJ, Ha YC, Koo KH. Five-year relative survival of patients with osteoporotic hip fracture. J Clin Endocrinol Metab 2014;9;9:97-100
10. Koot VC, Peeters PH, de Jong JR et al. Functional results after treatment of hip fracture: a multicentre, prospective study in 215 patients. Eur J Surg 2000;166:480-485
11. Brenneman SK, Barret-Connor E, Sajjan S et al. Impact of recent fracture on health-related quality of life in post-menopausal women. J Bone Miner Res 2006;21:809-816
12. Zimmerman S, Chandler JM, Hawkes W et al. Effect of fracture on the health care use of nursing home residents. Arch Intern Med 2002;162:1502-1508
13. Parikh S, Mogun H, Avorn J. Osteoporosis medication use in Nursing Home patients in with fractures in 1 US State. Arch Intern Med 2008;168:1111-1115
14. Gillespie LD, Gillespie WJ, Robertson MC et al. Interventions for preventing falls in elderly people. Cochrane Database Syst Rev CD000340, 2003
15. Parikh S, Brookhart MA, Stedman M et al. Correlations of nursing home characteristics with prescription of osteoporosis medications. Bone 2011;48:1164-1168
16. Colon-Emeric C, Lyles KW, Levine DA et al. Prevalence and predictors of osteoporosis treatment in nursing home residents with known osteoporosis or recent fracture. Osteoporos Int 2007;18:553-559
17. Wrigt RM. Use of osteoporosis mediations in older nursing facility residents. J Am Med Dir Assoc 2007;8:453-457
18. Lattanzio F, Mussi C, Scafato E et al. A computerized network on health care services for older people. J Nutr Health Aging 2010;14:238-242
19. Morris JN, Hawes C, Murphy K. Resident Assessment Instrument Training Manual and Resource Guide. Eliot Press Natick MA, 1991
20. SIGG, Società Italiana di Gerontologia e Geriatria, (accessed March 2016).
21. Morris JN, Fries BE, Morris SA. Scaling ADL’s within the MDS. J Gerontol 1999;4:M546-553
22. Morris JN, Fries BE, Mehr DR et al. MDS Cognitive Performance Scale. J Gerontol 1994;49:M174-182
23. World Health Organization. International Classification of Diseases. Ninth Revision, 1997
24. Parmelee PA, Thuras PD, Katz IR. Validation of the Cumulative Illness Rating Scale in a geriatric residential population. J Am Geriatr Soc 1995;43:130-137
25. Pahor M, Chrischilles EA, Guralnik JM, Brown SL, et al. Drug data coding and analysis in epidemiologic studies. Eur J Epidemiol 1994;10:405-411
26. Nguyen ND, Eisman JA, Nguyen TV. Anti-hip fracture efficacy of biphosphonates: a Bayesian analysis of clinical trials. J Bone Miner Res 2006;21:340-349.
27. Haasum Y, Fastbom J, Fratiglioni L. Undertreatment of osteoporosis in person with dementia? A population-base study. Osteoporos Int 2012;23:1061-1068
28. Weller I, Schatzker J. Hip fracture and Alzheimer’s disease in elderly institutionalized Canadians. Ann Epidemiol 2004;14:319-324
29. Kose N, Cuvalci S, Ekici G et al. The risk factors of fall and their correlation with balance, depression, cognitive impairment and mobility skills in elderly nursing home residents. Saudi Med J 2005;6:978-981
30. Duque G, Mallet L, Roberts A, et al. To treat or not to treat, that is the question: proceedings of the Quebec Symposium for the Treatment of Osteoporosis in Long-term Care Institutions, Saint-Hyacinthe, Quebec, November 5, 2004. J Am Med Dir Assoc 2006;7:435-441.
31. Cherubini A, Corsonello A, Lattanzio F. Polypharmacy in Nursing Home Residents: What Is the Way Forward? J Am Med Dir Assoc 2016;17:4-6.