C. Laffon de Mazières1,2, J.E. Morley3, C. Levy4, F. Agenes5, M. Barbagallo6, M. Cesari1,2, P. De Souto Barreto1,2, L.M. Donini7, J. Fitten8, A. Franco9, M. Izquierdo10, R.A. Kane11, F.C. Martin12, G. Onder13, J. Ouslander14, K. Pitkälä15, D. Saliba16,
A. Sinclair17, L. Rodriguez Manas18, B. Vellas1,2, Y. Rolland1,2
1. Department of Geriatric Medicine, Gérontopôle, Toulouse University Hospital (CHU de Toulouse), Toulouse, France; 2. Inserm UMR 1027, Toulouse, France; 3. Division of Geriatric Medicine, Saint Louis University School of Medicine, St Louis, MO; Division of Endocrinology, Saint Louis University School of Medicine, St Louis, MO; 4. Denver-Seattle Center of Innovation at the Denver VA Medical Center, Denver, CO; Division of Health Care Policy and Research, University of Colorado School of Medicine, Aurora, CO; 5. Office for Science and Technology, French Consulate in Los Angeles, Embassy of France in the United States, Los Angeles, CA, USA; 6. Geriatric Unit, University of Palermo, Palermo, Italy; 7. Sapienza University of Rome, Rome, Italy; 8. Psychiatry and Behavioral Sciences, David Geffen School of Medicine, University of California, Los Angeles, CA; Geriatric Psychiatry, Greater Los Angeles VA, Sepulveda Campus, Los Angeles, CA; 9. University of Nice-Sophia Antipolis, Nice, France; 10. Department of Health Sciences, Public University of Navarre, Navarra, Pamplona, Spain; 11. Center on Aging, Division of Health Policy & Management, School of Public Health, University of Minnesota, Minneapolis, Minnesota, USA; 12. King’s College London, London, UK; 13. Department of Geriatrics, Centro Medicina Invecchiamento, Università Cattolica del Sacro Cuore, Rome, Italy; 14. Charles E. Schmidt College of Medicine, Florida Atlantic University, Boca Raton, FL; Christine E. Lynn College of Nursing, Florida Atlantic University, Boca Raton, FL; 15. University of Helsinki, Department of General Practice and Primary Health Care, Helsinki University Hospital, Unit of Primary Health Care, Helsinki, Finland; 16. UCLA/JH Borun Center for Gerontological Research, University of California, Los Angeles, CA; Los Angeles Veterans Administration Geriatrics Research Education and Clinical Center, Los Angeles, CA; 17. Foundation for Diabetes Research in Older People, Diabetes Frail, Medici Medical Practice, Luton, UK; 18. Department of Geriatrics, Hospital Universitario de Getafe and School of Health Sciences, Universidad Europea de Madrid, Getafe, Spain. Corresponding author: Clarisse Laffon de Mazières, MD; Department of Geriatric Medicine, Gérontopôle, Toulouse University Hospital, 224 avenue de Casselardit, TSA 40031-31059 Toulouse cedex 9, France, Email: laffondemazieres.c@chu-toulouse.fr
Observers: S. Andrieu, L. Benattar, F. Bertin-Hugault, P. Denormandie, J. de Kerimel, P.-Y. de Kerimel, L. Dominguez, S. Guyonnet, F. Rey, S. Sasat, E. Tangalos, and H. Tissot.
Jour Nursing Home Res 2017;3:10-15
Published online February 1, 2017, http://dx.doi.org/10.14283/jnhrs.2017.2
Abstract
Institutionalization is generally a consequence of functional decline driven by physical limitations, cognitive impairments, and/or loss of social supports. At this stage, intervention to reverse functional losses is often too late. To be more effective, geriatric medicine must evolve to intervene at an earlier stage of the disability process. Could nursing homes (NHs) transform from settings in which many residents dwell to settings in which the NH residents and those living in neighboring communities benefit from staff expertise to enhance quality of life and maintain or slow functional decline? A task force of clinical researchers met in Toulouse on December 2, 2015, to address some of these challenges: how to prevent or slow functional decline and disabilities for NH residents and how NHs may promote the prevention of functional decline in community-dwelling frail elderly. The present article reports the main results of the Task Force discussions to generate a new paradigm.
Key words: Nursing home; prevention; functional decline; frailty.
Worldwide, the number of dependent older adults is projected to rise from 350 million in 2010 to 488 million in 2030 and 614 million in 2050 (1). These increases are expected to influence the percentage of older adults residing in nursing homes (NHs), which in 2010 in the United States represented approximately 9% of people aged 85 or older (2) and 6% in 2011 in France (3).
In 2011 in the United States, $134 billion was dedicated to institutional care (ie, skilled nursing facilities, nursing homes, and nursing facilities located in continuing care retirement communities) or about 31% of long-term services and supports expenditures with an additional $234 billion dedicated to informal care and $58 billion to community-based care (2). In 2012 in France, 8.3 billion euros were dedicated to health care for institutionalized people and increased each year (4). Similar increases are being observed throughout European nations.
Given rising costs in France, where 40% of dependent (defined as 2 or more activities of daily living [ADL] limitations) older adults are institutionalized (5), the National Assembly convened a panel of experts who estimated that an appropriate program organized in the community to prevent dependency would save a total of 10 billion euros in France (6). Such a recommendation to focus on prevention of disability has long been proposed as one potential strategy to respond to increasing levels of dependence and costs throughout the United States and Europe. Large randomized controlled trials have demonstrated an impact of primary prevention on morbidity (7). These observations mandate public health strategies and initiatives directed at prevention of disability (8). A shift toward prevention of disability would be consistent with trends in other medical disciplines such as oncology and cardiology that increasingly place an emphasis on proactive preventative approaches (eg, prevention of cancer or myocardial infarction) rather than reactive approaches after advanced disease has already developed.
Different pathways drive functional decline and development of disability with advanced age: loss of mobility, loss of cognitive function, loss of social support, iatrogenic events, and progression of disease. Earlier intervention to prevent decline and iatrogenic events may support geriatric medicine in meeting the challenge of slowing decline, and maintaining function and quality of life for as long as possible during aging.
Institutionalization is often a consequence of functional decline in older adults and is generally regarded as an outcome that signals failure of care systems to support elders effectively in the community (9-13). The expertise in NHs has long been questioned, but quality improvement is now reported in NH staff (eg, trained nurses and nurse aides, geriatricians, psychologists, dieticians, and physiotherapists). The NH staff could now shift NHs from a place of “dwelling” to a locus of activity both for current residents and for community-dwelling elderly at risk of functional loss (14). The goal would be to provide proactive, preventative approaches to delay disability and avoid iatrogenic events and loss of social supports modeled after other innovative community engagement initiatives such as the “Walk with a Doc” program that encourages physical activity by joining volunteer doctor in the community for a walk, many of which begin their walks at senior centers, assisted living facilities, and other locations where elders reside (15).
A Task Force of Experts in NH care and research met on December 2, 2015, in Toulouse, France, during the 2nd Nursing Home Research International Working Group (NHRIWG2015) conference with the support of the Office for Science and Technology of the Consulate General of France in Los Angeles, USA.
The aim of the workshop was to discuss intervention strategies to prevent disability and functional decline for NH residents. The second aim of the task force was to consider possible implementation strategies to prevent functional decline in community-dwelling frail elderly within the NH setting.
The Task Force was convened by the Global Aging Research Network (GARN), a network of the International Association of Gerontology and Geriatrics (IAGG).
How to Prevent Functional Decline and Disabilities for Nursing Home Residents?
Overview of the Nursing Home Health System in the United States
In the United States, 15,700 nursing homes cared for approximately 1.4 million residents on any given day (16) in 2012 but the number of nursing homes has been steadily decreasing compared to a decade earlier when there were more than 16,000 beds. NH care is still viewed as something to be avoided if possible and NH admission reduction has been actively sought. The declining number of nursing homes has been attributed to availability of assisted living facilities and long-term supports and services such as home and community-based services. Occupancy has also declined slightly over the past decade from 86% in 2003 to 83% in 2012. The majority of nursing homes (68%) are for-profit, whereas only 25% are nonprofit and 7% are government-owned; the majority serve between 26 and 100 residents and approximately one-third serve more than 100 residents. NH staff are usually trained, but available time dedicated to resident care is limited. The vast majority of direct care is provided by nurse aides, who represent 65% of the total nursing full-time equivalents (FTEs), with an average of 2.46 hours of care per resident each day compared with 23% FTE and 0.85 hours of medical care for licensed practical nurses and only 12% FTE and 0.52 hours of medical care for registered nurses in nursing homes. Nearly all nursing home residents in the United States need assistance with bathing, dressing, and toileting, and more than half also require assistance with eating.
Overview of the Nursing Home Health System in Europe
Over the past 10 years, the growing number of patients who benefit from long-term care at home, especially in Sweden, France, and United Kingdom, generated a larger proportion of severely disabled residents living in NHs (17). In Europe, as in other regions of the world, NHs are more unique than similar (18). Since 2000, the number of NH beds has been increasing in Europe. In 2013 in Belgium, there were 72.1 NH beds per 1000 population aged 65 and older (59 in France, 18.9 in Italy) (19). Similar trends of increasing numbers of long-term care beds were observed in Finland or Iceland between 2000 and 2013. During that same period, Spain reported one of the highest increase of NH beds by adding an average of 3.7 beds per 1000 population older 65 than years, each year in institutions (19).
The Paradox of Nursing Home Funding
Although it was beyond the scope of our Task Force to comment on the complexities of NH funding, all countries face similar challenges of increasing costs related to financing the care of older adults and incentives that do not necessarily align with prevention of disability. In the United States, the majority of funding for nursing home care is provided by 3 main sources (the first 2 are federally funded): (1) Medicaid (63%), for those who do not have personal funds to pay for care, (2) Medicare (14%), primarily for persons undergoing post-acute rehabilitation, and (3) private out-of-pocket payments (22%) (20). The median annual cost for nursing home care with a semi-private room in 2012 was US $73,000 per year or approximately US $200 daily (21). This cost exceeds the median household income of older persons several-fold and frequently results in the exhaustion of personal assets and reliance on the federally funded Medicaid program to cover the cost of care. From a policy perspective, this is significant because there is now financial and social pressure causing a trend toward reductions in nursing home expenditures or bed as more emphasis is placed on home- and community-based services. Medicaid expenditures on institutional care decreased from 76% in 1997 to 52% in 2011 as home and community-based services rose from 24% to 48% of Medicaid expenditures during this 15-year time period (22). Rehabilitation services in NHs for older adults that tend to be low intensity (23) do not appear to adequately address deficits in physical function or performance and tend to foster inactivity (24). Both low-intensity interventions and inactivity may perpetuate further functional decline or impede maximal recovery. This observation is concerning because decline in ability to perform ADL and subsequent increased need for caregiver assistance are strongly associated with the need for costly long-term services because in most developed nations, the size of NH payment depends on the residents’ degree of disability.
In France, the model of funding for NH in France relies on 2 comprehensive assessment tools [the GIR (Groupe Iso-Ressource) (25) and the PATHOS (26)] that are regularly performed by staff members. Briefly, the more disabled and ill residents generate more money for the institution. For policy makers, the way NHs are financed is relevant, but for NH staff this relationship between commitment to residents and resources can create perverse incentives and may not encourage the medical director, the registered nurse, or the administrator to implement preventive measures in their care facility. Prevention of adverse events, such as falls, hospitalization, infection, iatrogenic events, undernutrition, wounds, and finally functional decline is however regarded by NH staff as the main driver for quality improvement and for core health care. Prevention of these adverse events is also a clinically meaningful axis of care to improve the quality of life of the residents.
In summary therefore, on one hand carers are encouraged to improve care quality and to reduce the risk of functional decline, but on the other hand, financial incentives are such that resources are greater when functional status is worse. Based on these observations, Task Force members recommend reframing NH care with the purpose of addressing the lack of rigor in rehabilitation, which currently results in failure to sustain or slow the loss of function. Policies to promote reframing of the focus in NHs need to be in alignment with the highest practicable level of care.
Changing the Culture in the Nursing Home
Nursing home residents are at risk of falls (27), malnutrition (28), weight loss (29), pressure ulcers (30), restraints (31, 32) polypharmacy, and inappropriate drug prescribing (33). In fact, some NHs themselves may be a risk factor for the residents. For instance, previous studies showed that in addition to resident characteristics, NH characteristics were associated with potentially inappropriate drug prescribing (34), particularly for neuroleptic drugs (35). Potentially avoidable hospitalizations are another high risk for NH residents. Hospitalizations are often associated with added disabilities due to iatrogenic events (36-38).
Workforce issues also are important when considering the role of prevention. In a survey of French NH staff, researchers found that between completing the broad range of everyday tasks necessary to support basic daily living and managing behavioral disturbances associated with dementia, staff report little time for promoting prevention programs (39). Prevention of functional decline was ranked eighth in terms of importance according to coordinating physicians and staff in the NH setting (39).
In France, the mean age of NH residents is 85 years and more than half of this population is bedridden or very dependent (39, 40), requiring much commitment on behalf of nurses and nurse aides, and evidence suggests that this disability accelerates following NH admission. In the IQUARE study, an 18-month multicenter controlled trial, one-third of the NH residents reported a low level of disability (ADL score of 4 or higher) at baseline (40). However, after a short 18-month period, 30% of residents had a lower ADL score (Figure 1) (41). Although these results may represent an unavoidable decline, interventions to prevent or slow decline are needed.
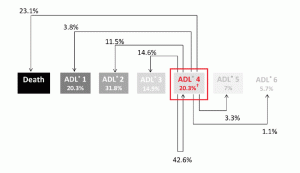
Figure 1
Functional decline at 18 months in NH residents according to the baseline ADL* score. Personal data IQUARE
(175 EHPAD; 6275 residents)
*A total score of 6 indicates full function, and 0 indicates severe disability. †The percentages under the ADL score correspond to the distribution of residents at baseline.
The Task Force recognized that there is an urgent need for cultural change to incorporate physical activity as an integral intervention as it is one of the most important components in improving the functional capacity of frail older people living in NHs (42, 43). With many residents spending at least 17 hours in bed daily (44), encouraging mobility, if possible, should be an important objective in each resident’s plan of care. Physical activity is a key factor contributing to maintenance of muscle mass and functional status (ie, leading to positive effects of exercise tolerance, falls reduction, balance, cardiorespiratory fitness, and strength performance), especially when multiple physical conditioning components (ie, strength, endurance, and balance) are included in the exercise intervention compared with only one type of exercise (42, 43). Multicomponent exercise programs, and particularly those including strength training, are the most effective interventions to delay disability and other adverse events (42). Indeed, it has been recently reported that multicomponent exercise training including explosive resistance training improved neuromuscular function and functional outcomes in frail NH nonagenarians after long-term physical restraint (43), as well as in frail multimorbid patients (45). Furthermore, physical exercise administration is relatively free of potential unwanted side effects.
Can Nursing Home Staff Provide Expertise in the Prevention of Functional Decline for Older People Living in the Community?
Screening Frailty and Cognitive Decline in the Elderly
Frailty is usually defined as a state of growing vulnerability that increases the risk of adverse outcomes, resulting in worsening physical function (46). Preventive strategies, aimed at reducing the burden of frailty in NH residents and in the community, necessarily imply a careful and objective evaluation of the patient’s needs and resources. The early stage of disability is occasionally reversible, and screening for frailty is conducive to preventive interventions (47). Various international initiatives have been proposed to screen and implement prevention programs for frail older adults living in the community (48).
The Toulouse Gérontopôle Frailty Clinic serves as a specialized team located in a day hospital who assess, manage, and prevent disability in frail older adults (48-50). Could this team be replicated for dissemination into community NHs such that resources were in closer proximity to the frail older adult’s residence? Nursing homes have many of the assets to participate in frailty assessment and become multidisciplinary outpatient care centers for the benefit of the community (51).
The Necessity of a Multidomain Intervention
The complex health status of frail elders requires a multidomain and interprofessional approach to prevent or slow functional decline. Multidomain interventions, designed by a “simple addition” of strategies against multiple risk factors/diseases, may result in unfavorable cost-effectiveness, infeasibility, and harm (52, 53). Interventions should be tailored for each individual according to personal needs and system resources. Multidomain interventions aimed at targeting multiple risk factors (eg, poor cognitive function and sedentary lifestyle) (54, 55) coordinated by geriatricians have been shown to make a positive impact on older persons (56, 57). Recent clinical trials have been designed to test combinations of interventions aimed at reducing functional decline (58-61), and have shown that multidomain intervention can reduce decline (61-64). Although geriatricians are in short supply in most nations, the Task Force posited that principles from these clinical trials could be taught to community-based aging specialists for dissemination to NHs interested in focusing on prevention both within the NH and potentially serving as a resource to community-dwelling elders in their community.
Can Nursing Homes Implement a Multidomain Intervention for Frail Older People Living in the Community?
Recognizing the many challenges in NHs with staff turnover and maintaining quality of care, NHs have many important assets. NHs provide a wide range of geriatrics experts depending on the country (geriatrician, general practitioner, pharmacist, psychologist, nurses, dietician, physiotherapist, and occupational therapist) dedicated to care of older patients. The Task Force speculated that in some countries, frail community-dwelling older people could have access to these specialists to obtain individual recommendations. It suggested that the “Walk with a Doc” program be adapted to create a program “Function at the Facility” in which community-dwelling elders are invited to a “function” at the nursing facility once a month to meet the staff and ask questions about staying active.
Another important asset is that NHs are located in communities where community-dwelling older adults are also located. In some locations, NHs also have space, equipment, and organization that could be used to arrange specific interventions such as physical, social, or cognitive activities.
Finally, in some countries, NHs could be adapted to implement multifactorial approaches to reduce functional decline. An important characteristic of efficient intervention for frail elderly living in the community is the involvement of the primary care physician (65). Primary care professionals in certain countries such as France are very involved in NHs, and general practitioners have strong collaborations with these facilities. With his or her weekly visits and the insight on patients’ health and functional and psychological status, the general practitioner establishes close relationships with NH staff, thus allowing personally designed care. This on-site organization is adapted to NH residents and could easily be extended to the frail elderly people living in the community. Beyond this strategy, the staff’s mission will promote a favorable new image of the NH.
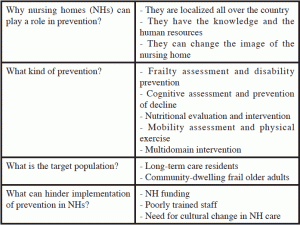
In summary, the Task Force group proposes that NHs serve as models of care focused on maintenance of function and slowing of decline among both NH and community-dwelling frail elders. NH staff could ideally provide comprehensive geriatric assessment and invite frail elders to the NH followed by creation of a multidomain intervention tailored to local technical and human resources (Table 1).
Conclusions and Perspectives
Geriatric medicine and NHs must evolve to a paradigm of proactive prevention rather than remain in a paradigm of reactive repair. New strategies are needed to tackle disability upstream. Evidence suggests that rehabilitative efforts in NHs are inadequate and residents would benefit from active preventive measures to avoid functional decline. In an ideal state, NH facilities would tap into current assets of geriatric expertise and capital within the community to focus on prevention of functional decline. Clearly, this reframing would require fundamental changes to the existing structure, but the demographic imperative will likely demand such transformation.
This article is a co-publication of JAMDA – The Journal of Post-Acute and Long-Term Care Medicine and the Journal of Nursing Home Research.
This article was published in JAMDA – The Journal of Post-Acute and Long-Term Care Medicine. Laffon de Mazières C, Morley JE, Levy C, et al. Prevention of Functional Decline by Reframing the Role of Nursing Homes? J Am Med Dir Assoc 2016;18:105–110. © 2016 AMDA – The Society for Post-Acute and Long-Term Care Medicine.
Acknowledgments: We thank Constance de Seynes for her valuable work.
Funding: This work was supported by an educational grant from the Office for Science and Technology of the Consulate General of France in Los Angeles, USA.
Conflict of interest: The authors declare no conflicts of interest.
References
1. Prevalence of dependence. World Alzheimer Report 2013. Available at: http://www.alz.co.uk/research/WorldAlzheimerReport2013.pdf. Accessed March 6, 2016.
2. Congressional Budget Office. Rising demand for long-term services and supports for elderly people. Available at: https://www.cbo.gov/sites/default/files/113th-congress-2013-2014/reports/44363-LTC.pdf. Accessed March 6, 2016.
3. Direction de la recherche, des études, de l’évaluation et des statistiques (DREES). L’offre en établissements d’hébergement pour personnes âgées en 2011. Available at: http://drees.social-sante.gouv.fr/IMG/pdf/er877.pdf. 2014. Accessed April 30, 2016.
4. INSEE. Tableaux de l’Économie Française—Édition 2014–février 2014. Available at: http://www.insee.fr/fr/ffc/tef/tef2014/T14F096/T14F096.pdf. Accessed March 6, 2016.
5. Direction de la recherche, des études, de l’évaluation et des statistiques (DRESS). Projection des populations âgées dépendantes. Available at: http://drees.social-sante.gouv.fr/IMG/pdf/article43.pdf. Accessed March 6, 2016.
6. Assemblée des départements de France (ADF). 55 propositions de l’ADF pour relever le défi de la perte d’autonomie. mai 2011. Available at: http://www.aidautonomie.fr/IMG/pdf/20110523_ADF-perte-autonomie-55-propositions-mai-2011.pdf. Accessed March 6, 2016.
7. Fries JF. The compression of morbidity: Near or far? Milbank Q 1989;67:208–232.
8. Fries JF. Strategies for reduction of morbidity. Am J Clin Nutr 1992;55:1257S–1262S.
9. Bakker C, de Vugt ME, van Vliet D, et al. Predictors of the time to institutionalization in young- versus late-onset dementia: Results from the Needs in Young Onset Dementia (NeedYD) study. J Am Med Dir Assoc 2013;14:248–253.
10. Brodaty H, Connors MH, Xu J, et al. PRIME study group. Predictors of institutionalization in dementia: A three year longitudinal study. J Alzheimers Dis 2014;40:221–226.
11. Hajek A, Brettschneider C, Lange C, et al. Longitudinal predictors of institutionalization in old age. PLoS One 2015;10:e0144203.
12. Martinez-Reig M, Ahmad L, Duque G. The orthogeriatrics model of care: Systematic review of predictors of institutionalization and mortality in post-hip fracture patients and evidence for interventions. J Am Med Dir Assoc 2012;13:770–777.
13. Stineman MG, Xie D, Streim JE, et al. Home accessibility, living circumstances, stage of activity limitation, and nursing home use. Arch Phys Med Rehabil 2012;93:1609–1616.
14. Morley JE. Clinical practice in nursing homes as a key for progress. J Nutr Health Aging 2010;14:586–593.
15. Walk with a Doc—Just a walk in the park. Available at: http://walkwithadoc.org/. Accessed November 16, 2016.
16. Harris-Kojetin L, Sengupta M, Park-Lee E, et al. Long-term care services in the United States: 2013 overview. Vital Health Stat 2013;3:1–107.
17. Organisation for Economic Co-operation and Development (OECD). Recipients of long-term care. In: Health at a Glance 2015. Paris: OECD; 2015:200–201. Available at: http://www.oecd-ilibrary.org/content/chapter/health_glance-2015-75-en. Accessed September 2, 2016.
18. Sanford AM, Orrell M, Tolson D, et al. An international definition for “nursing home”. J Am Med Dir Assoc 2015;16:181–184.
19. Organisation for Economic Co-operation and Development (OECD). Health at a Glance 2015. Available at: http://www.oecd-ilibrary.org/content/book/health_glance-2015-en. Accessed March 8, 2016.
20. Harrington C, Carrillo H, Dowdell M, et al. Nursing facilities, staffing, residents and facility deficiencies, 2005 through 2010. Department of Social and Behavioral Sciences, University of California, San Francisco. Available at: http://www.pascenter.org. Accessed April 30, 2016.
21. The Scan Foundation. who_pays_for_ltc_us_jan_2013_fs.pdf. Available at: http://www.thescanfoundation.org/sites/default/files/who_pays_for_ltc_us_jan_2013_fs.pdf. 2013. Accessed April 30, 2016.
22. Martin AB, Hartman M, Whittle L, Catlin A. Team the NHEA. National health spending in 2012: Rate of health spending growth remained low for the fourth consecutive year. Health Aff (Millwood) 2014;33:67–77.
23. Mangione KK, Lopopolo RB, Neff NP, et al. Interventions used by physical therapists in home care for people after hip fracture. Phys Ther 2008;88:199–210.
24. Grant PM, Granat MH, Thow MK, Maclaren WM. Analyzing free-living physical activity of older adults in different environments using body-worn activity monitors. J Aging Phys Act 2010;18:171–184.
25. Code de L’action Sociale et Des Familles – Article R314-170-6. Vol R314-170-6.
26. Code de L’action Sociale et Des Familles – Article R314-170-4. Vol R314-170-4.
27. Rubenstein LZ, Josephson KR, Robbins AS. Falls in the nursing home. Ann Intern Med 1994;121:442–451.
28. Verbrugghe M, Beeckman D, Van Hecke A, et al. Malnutrition and associated factors in nursing home residents: A cross-sectional, multi-centre study. Clin Nutr 2013;32:438–443.
29. Wirth R, Streicher M, Smoliner C, et al. The impact of weight loss and low BMI on mortality of nursing home residents—Results from the nutritionDay in nursing homes. Clin Nutr 2016;35:900–906.
30. Khor HM, Tan J, Saedon NI, et al. Determinants of mortality among older adults with pressure ulcers. Arch Gerontol Geriatr 2014;59:536–541.
31. Hofmann H, Schorro E, Haastert B, Meyer G. Use of physical restraints in nursing homes: A multicentre cross-sectional study. BMC Geriatr 2015;15:129.
32. Tolson D, Morley JE. Physical restraints: Abusive and harmful. J Am Med Dir Assoc 2012;13:311–313.
33. Morley JE. Inappropriate drug prescribing and polypharmacy are major causes of poor outcomes in long-term care. J Am Med Dir Assoc 2014;15:780–782.
34. Cool C, Cestac P, Laborde C, et al. Potentially inappropriate drug prescribing and associated factors in nursing homes. J Am Med Dir Assoc 2014;15:850.e1–850.e9.
35. Laffon de Mazières C, Lapeyre-Mestre M, Vellas B, et al. Organizational factors associated with inappropriate neuroleptic drug prescribing in nursing homes: A multilevel approach. J Am Med Dir Assoc 2015;16:590–597.
36. Ouslander JG, Schnelle JF, Han J. Is this really an emergency? Reducing potentially preventable emergency department visits among nursing home residents. J Am Med Dir Assoc 2015;16:354–357.
37. Burke RE, Rooks SP, Levy C, et al. Identifying potentially preventable emergency department visits by nursing home residents in the United States. J Am Med Dir Assoc 2015;16:395–399.
38. Sourdet S, Lafont C, Rolland Y, et al. Preventable iatrogenic disability in elderly patients during hospitalization. J Am Med Dir Assoc 2015;16:674–681.
39. Rolland Y, Abellan van Kan G, Hermabessiere S, et al. Descriptive study of nursing home residents from the REHPA network. J Nutr Health Aging 2009;13:679–683.
40. de Souto Barreto P, Lapeyre-Mestre M, Mathieu C, et al. A multicentric individually-tailored controlled trial of education and professional support to nursing home staff: Research protocol and baseline data of the IQUARE study. J Nutr Health Aging 2013;17:173–178.
41. Rolland Y, Mathieu C, Piau C, et al. Improving the quality of care of long-stay nursing home residents in France. J Am Geriatr Soc 2016;64:193–199.
42. Cadore E, Casas-Herrero A, Millor N, et al. Multicomponent exercises including muscle power training enhance muscle mass, power output, and functional outcomes in institutionalized frail nonagenarians. Age (Dordr) 2014;36:773–785.
43. Cadore E, Moneo A, Mensat M, et al. Positive effects of resistance training in frail elderly patients with dementia after long-term physical restraint. Age (Dordr) 2014;36:801–811.
44. Bates-Jensen BM, Alessi CA, Cadogan M, et al. The Minimum Data Set bedfast quality indicator: Differences among nursing homes. Nurs Res 2004;53:260–272.
45. Cadore E, Izquierdo M. Exercise interventions in polypathological aging patients that coexist with diabetes mellitus: Improving functional status and quality of life. Age (Dordr) 2015;37:64.
46. Clegg A, Young J, Iliffe S, et al. Frailty in elderly people. Lancet Lond Engl 2013;381:752–762.
47. Gill TM, Gahbauer EA, Allore HG, Han L. Transitions between frailty states among community-living older persons. Arch Intern Med 2006;166:418–423.
48. Tavassoli N, Guyonnet S, Abellan Van Kan G, et al. Description of 1,108 older patients referred by their physician to the “Geriatric Frailty Clinic (G.F.C.) for Assessment of Frailty and Prevention of Disability” at the gerontopole. J Nutr Health Aging 2014;18:457–464.
49. Morley JE, Adams EV. Rapid geriatric assessment. J Am Med Dir Assoc 2015;16:808–812.
50. Kera T, Kawai H, Yoshida H, et al. Classification of frailty using the Kihon checklist: A cluster analysis of older adults in urban areas. Geriatr Gerontol Int 2016. http://dx.doi.org/10.1111/ggi.12676 [Epub ahead of print].
51. Morley JE, Morris JC, Berg-Weger M, et al. Brain health: The importance of recognizing cognitive impairment: An IAGG consensus conference. J Am Med Dir Assoc 2015;16:731–739.
52. Ferrucci L, Windham BG, Fried LP. Frailty in older persons. Genus 2005;61:39–53.
53. World Health Organization. World report on ageing and health. Available at: http://apps.who.int/iris/bitstream/10665/186463/1/9789240694811_eng.pdf?ua=1. Accessed March 11, 2016.
54. Loraine J, Taylor S, McAllister M. Cognitive and physical stimulation therapy. J Am Med Dir Assoc 2014;15:140–141.
55. Berg-Weger M, Tebb S, Henderson-Kalb J, et al. Cognitive stimulation therapy: A tool for your practice with persons with dementia? J Am Med Dir Assoc 2015;16:795–796.
56. Lisk R, Yeong K, Nasim A, et al. Geriatrician input into nursing homes reduces emergency hospital admissions. Arch Gerontol Geriatr 2012;55:331–337.
57. Ellis G, Whitehead MA, Robinson D, et al. Comprehensive geriatric assessment for older adults admitted to hospital: Meta-analysis of randomised controlled trials. BMJ 2011;343:d6553.
58. Cesari M, Demougeot L, Boccalon H, et al. The Multidomain Intervention to preveNt disability in ElDers (MINDED) project: Rationale and study design of a pilot study. Contemp Clin Trials 2014;38:145–154.
59. Ngandu T, Lehtisalo J, Solomon A, et al. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): A randomised controlled trial. Lancet Lond Engl 2015;385:2255–2263.
60. Vellas B, Carrie I, Gillette-Guyonnet S, et al. MAPT study: A Multidomain Approach for Preventing Alzheimer’s disease: Design and baseline data. J Prev Alzheimers Dis 2014;1:13–22.
61. Tarazona-Santabalbina FJ, Gómez-Cabrera MC, Pérez-Ros P, et al. A multicomponent exercise intervention that reverses frailty and improves cognition, emotion, and social networking in the community-dwelling frail elderly: A randomized clinical trial. J Am Med Dir Assoc 2016;1:426–433.
62. Ng TP, Feng L, Nyunt MSZ, et al. Nutritional, physical, cognitive, and combination interventions and frailty reversal among older adults: A randomized controlled trial. Am J Med 2015;128:1225–1236.e1.
63. Abizanda P, López MD, García VP, et al. Effects of an oral nutritional supplementation plus physical exercise intervention on the physical function, nutritional status, and quality of life in frail institutionalized older adults: The ACTIVNES study. J Am Med Dir Assoc 2015;16:439.e9–439.e16.
64. Singh NA, Quine S, Clemson LM, et al. Effects of high-intensity progressive resistance training and targeted multidisciplinary treatment of frailty on mortality and nursing home admissions after hip fracture: A randomized controlled trial. J Am Med Dir Assoc 2012;13:24–30.
65. Luijks H, Lucassen P, van Weel C, et al. How GPs value guidelines applied to patients with multimorbidity: A qualitative study. BMJ Open 2015;5:e007905.